|
8/16/2023 0 Comments Oxidation chart periodic table
Many of these oxides dissolve in water to give the oxo anion in which the element has the same oxidation number as the oxide. Many of the covalent oxides of the very acidic cations (hypothetical) exhibit acidic properties. Although these oxides do not significantly alter the pH of the water, they are still bases and neutralize strong acids. These oxides do not introduce significant amounts of O 2- ion into the solution so hydroxide ions are not produced. The oxides of weakly acidic cations and moderately acidic cations are insoluble in water. The hydroxides of feebly acidic cations are not deliquescent. The oxides of feebly acidic cations react exothermically with water producing the hydroxide. Lime can be prepared comercially by the thermal decomposition of limestone. The oxides of feebly acidic cations are more common. These oxides cannot be prepared by dehydrating the hydroxide at high temperature. The oxides of nonacidic cations are so water reactive that they are seldom seen. Metal oxides which exhibit this behavior are termed basic oxides because they act as bases. These aqueous solutions are strongly basic. The dissolution is highly exothermic generating the hydroxide of the cation. This is an example the leveling property of very strong bases.Īs predicted by the generalized solubility rules, only those metal oxides of the nonacidic and feebly acidic cation dissolve in water. 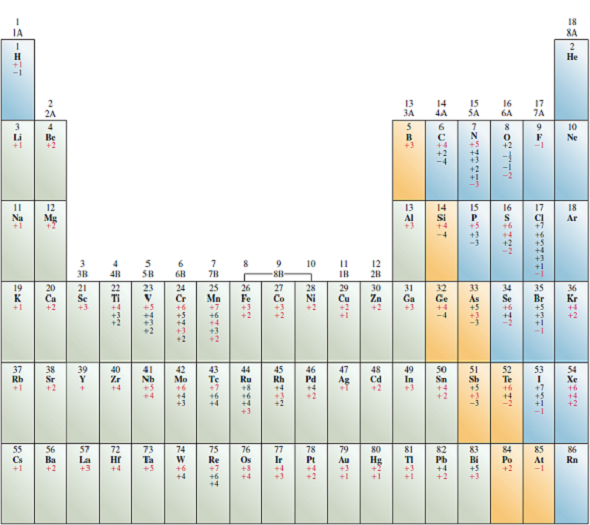
Since hydroxide ion is the strongest base that can persist in water, the oxide ion reacts quantitatively with water to generate hydroxide ions. The oxide ion is a very strongly basic anion due to its very small size and high charge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
